 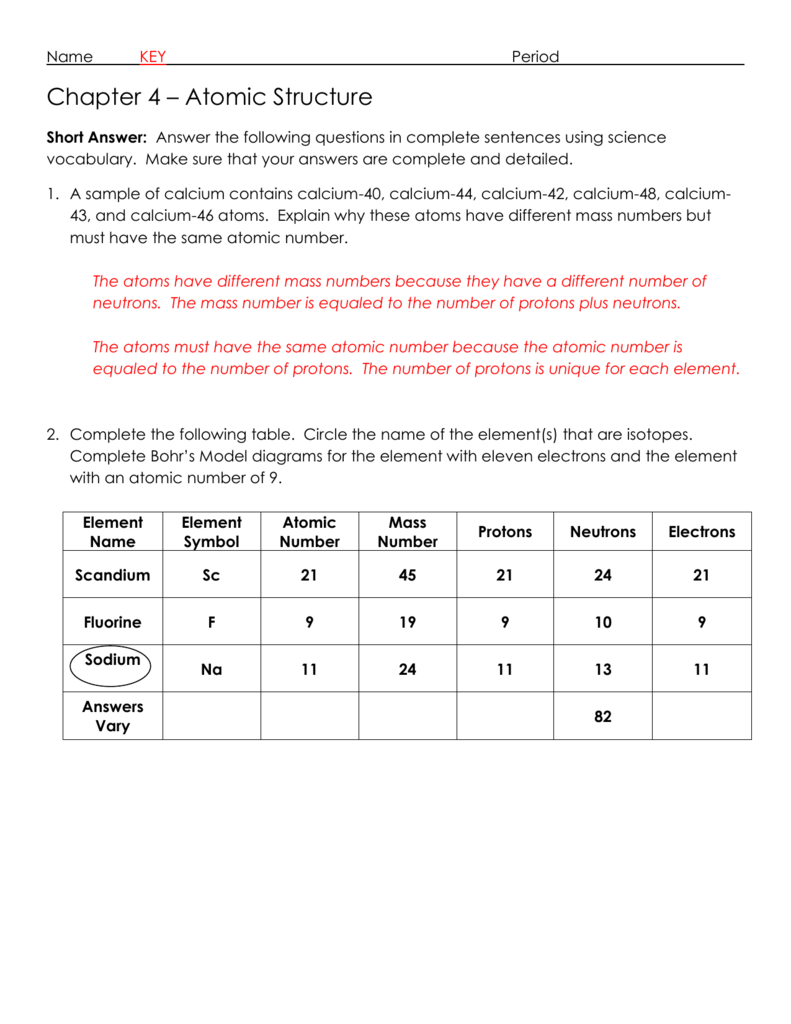
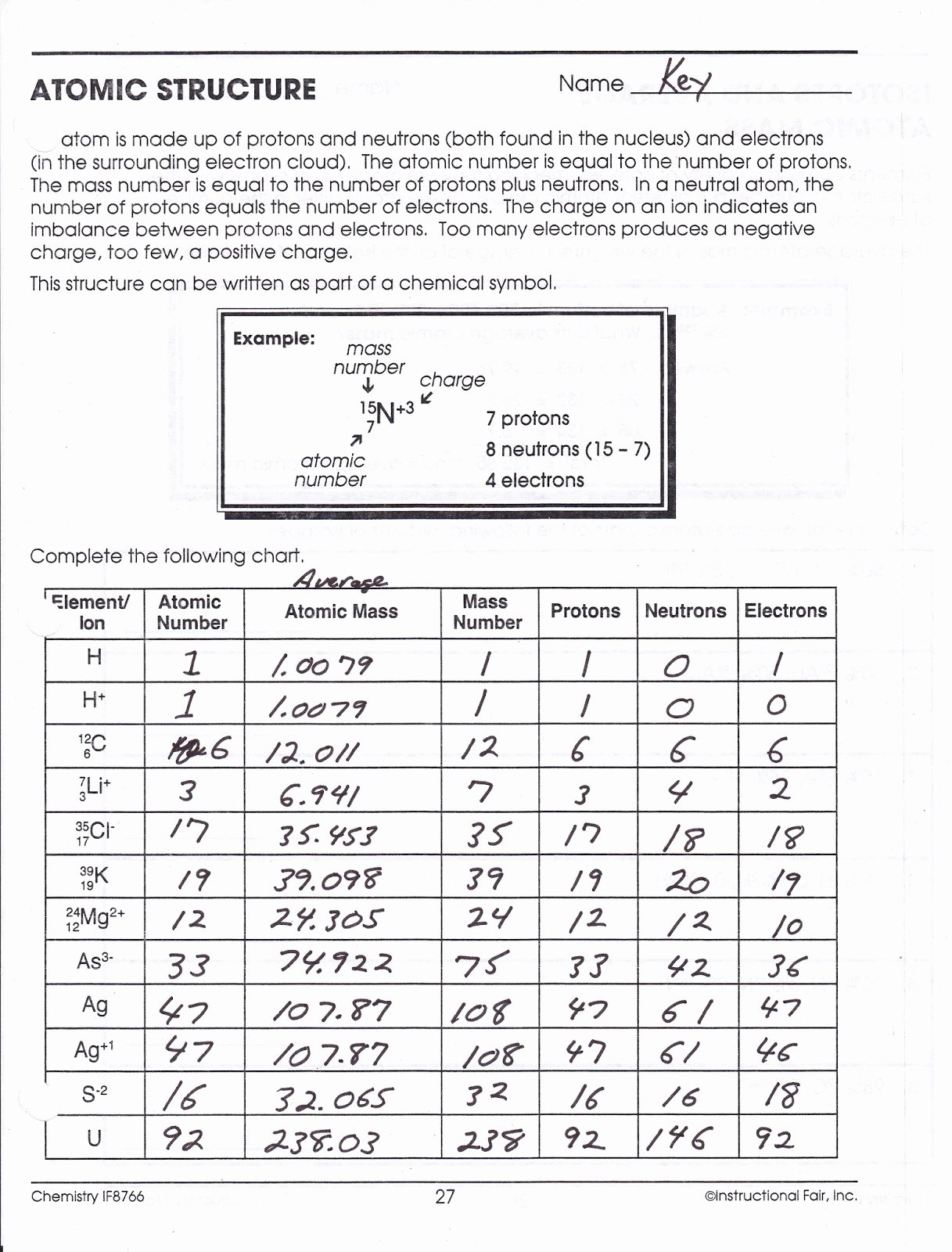
1.1.4 define mass number as the total number of protons and neutrons in an atom.1.1.3 define atomic number as the number of protons in an atom.1.1.1 describe the structure of an atom as a central positively charged nucleus containing protons and neutrons (most of the mass) surrounded by orbiting electrons in shells.Unit C1: Structures, Trends, Chemical Reactions, Quantitative Chemistry and Analysis.1.1.7 calculate the number of protons, neutrons and electrons in an atom or an ion and deduce the charge on an ion or determine the number of subatomic particles given the charge.1.1.5 define mass number as the total number of protons and neutrons in an atom.1.1.4 define atomic number as the number of protons in an atom. 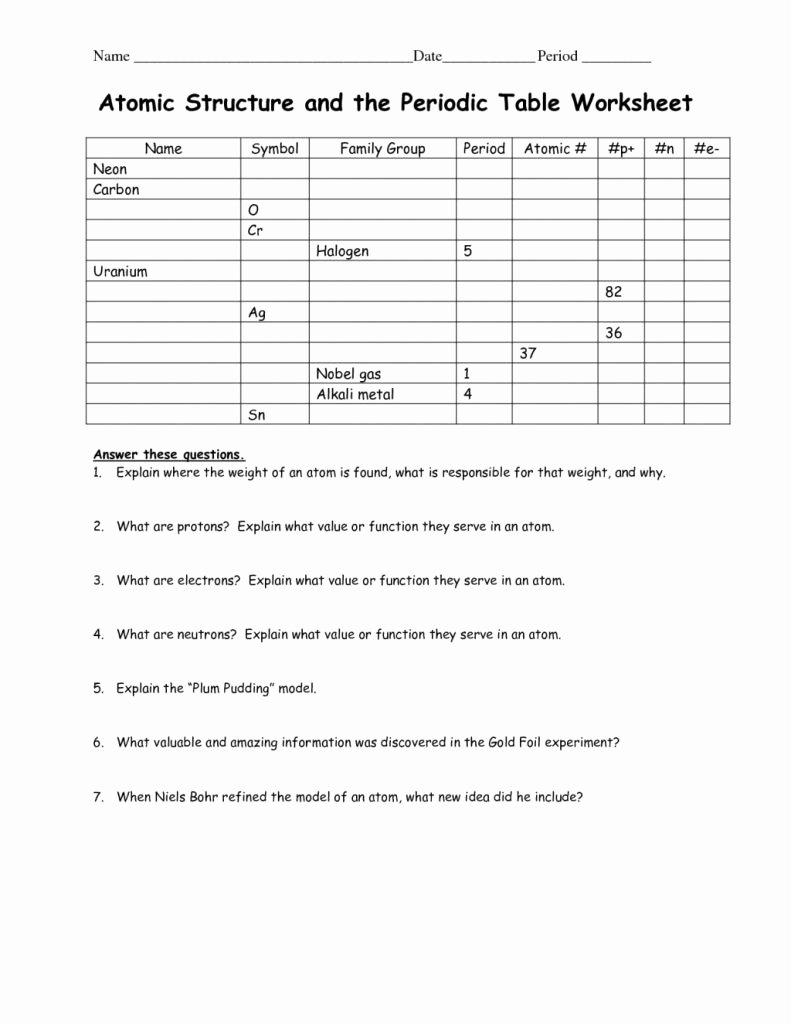
1.1.2 describe the structure of an atom as a central positively charged nucleus containing protons and neutrons (most of the mass) surrounded by orbiting electrons in shells.Unit 1: Structures, Trends, Chemical Reactions, Quantitative Chemistry and Analysis. 
The outcome for each group is externally validated by the teacher. The focus is on the whole group rather than individual understanding with peer mentoring a feature of this activity. Initial self assessment is followed by peer questions and judgements. The initial image provides a catalyst for sharing the criteria related to the learning objectives.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
